Correct Answer

verified
Correct Answer
verified
Essay
Propose an efficient synthesis of 1-butene from propyne:
Correct Answer

verified
Correct Answer
verified
Essay
Devise an efficient synthesis of the diol shown below starting with 1,1,3,3-tetramethyl-2-ethylcyclohexane. 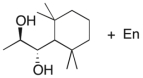
Correct Answer

verified
 Alternatively steps...
Alternatively steps...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
For the transformation shown, select the most appropriate reagent(s) to effect the change. 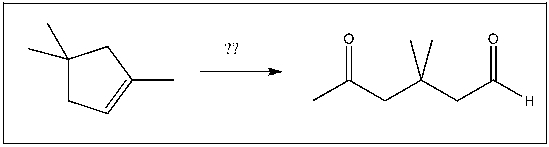
A) 1) OsO4; 2) NaHSO3, H2O
B) 1) Hg(OAc) 2, H2O; 2) NaBH4
C) 1) RCO3H; 2) H3O+
D) H2SO4, H2O
E) 1) O3; 2) DMS
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Propose a strategy to prepare the target as one of the major products: 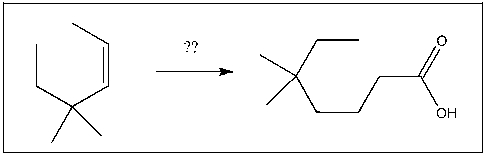
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major product(s) for the following reaction: 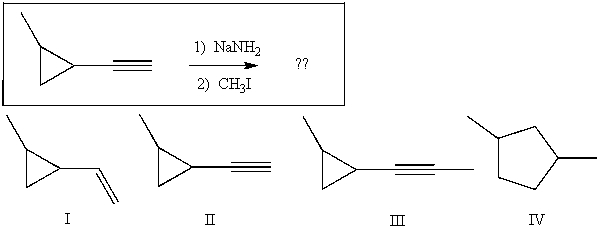
A) I
B) II
C) III
D) IV
E) II and III
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Reaction of compound A (molecular formula = C7H12) with sodium amide followed by reaction with 1-bromobutane produces (CH3) 2CHCH(CH3) CC(CH2) 3CH3. What is the name of compound A?
A) 2,3-dimethylnon-4-yne
B) 2,2-dimethylpent-1-yne
C) 3,4-dimethylpent-1-yne
D) 4,4-dimethylhept-1-yne
E) 1-heptyne
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the transformation shown, select the most appropriate reagent(s) to effect the change. 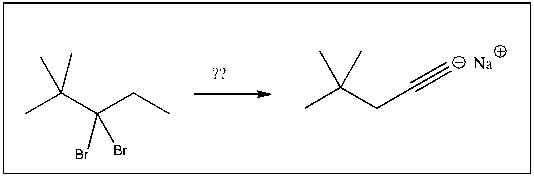
A) 1) OsO4; 2) NaHSO3, H2O
B) H2SO4, H2O, HgSO4
C) NaOH
D) xs NaNH2
E) 1) O3; 2) DMS
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Devise a method of converting acetylene into the polymer shown below. 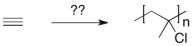
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the best reagent to convert 4,5-dimethylhex-2-yne to 4,5-dimethylhex-1-yne.
A) 1) OsO4; 2) NaHSO3, H2O
B) HBr, ROOR
C) NaBr
D) 1) xs NaNH2; 2) H3O+
E) HBr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Propose an efficient method of completing the following transformation: 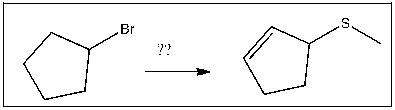
Correct Answer

verified
Correct Answer
verified
Essay
Devise an efficient synthesis of the diol shown below starting with 1,1,3,3-tetramethyl-2-ethylcyclohexane. 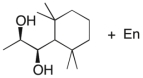
Correct Answer

verified
 Alternatively steps...
Alternatively steps...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Starting with a primary alkyl bromide, which of the following results in an overall increase in the length of the carbon skeleton by one carbon?
A) substitute bromide with acetylide, then cleave the triple bond.
B) substitute bromide with acetylide, then reduce the alkyne to an alkene.
C) substitute bromide with methoxide.
D) eliminate hydrogen bromide to produce an alkene.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Propose a multi-step synthetic sequence to accomplish the transformation below. 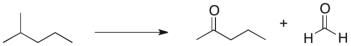
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using retrosynthetic analysis, determine which compound(s) could lead to the alkane shown below in a single step: 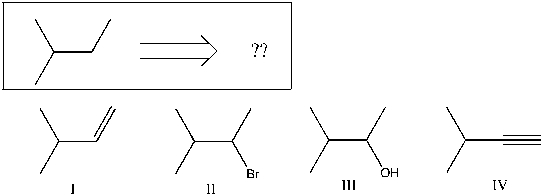
A) II or III
B) I or IV
C) I
D) III
E) IV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 95 of 95
Related Exams