A) Yes, it is 30 mM.
B) Yes, it is 30 mM/sec.
C) Yes, it is 60 mM/sec
D) Yes, it is 60 mM
E) No this data does not follow Michaelis-Menten kinetics
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The KM can be considered to be the same as the dissociation constant KS for E + S binding if
A) the concentration of [ES] is unchanged.
B) ES E + P is fast compared to ES E + S.
C) k1 >> k2
D) k2 << k-1.
E) this statement cannot be completed because KM can never approximate KS.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The catalytic efficiency of an enzyme can never exceed
A) k2.
B) k1.
C) k-1.
D) k-1 + k2.
E) (k-1 + k2) /k1.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Matching -The E+S E+P reaction is ______.
A) isozymes
B) [A]
C) the rate constant
D) Ping Pong
E) bimolecular
F) ES complex
G) random ordered
H) competitive inhibition
I) unimolecular
J) [A]²
K) Competitive inhibition
L) phosphorylation
M) small KS
N) large KS
O) uncompetitive inhibition
P) [B]
R) D) and O)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Reaction that is first order with respect to A and B
A) is dependent on the concentration of A and B.
B) is dependent on the concentration of A.
C) has smaller rate constants than first-order reactions regardless of reactant concentration.
D) is independent of reactant concentration.
E) is always faster than first-order reactions due to loss of concentration dependence.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Matching -A common type of covalent modification of regulatory enzymes involves ______ of serine residues.
A) isozymes
B) [A]
C) the rate constant
D) Ping Pong
E) bimolecular
F) ES complex
G) random ordered
H) competitive inhibition
I) unimolecular
J) [A]²
K) Competitive inhibition
L) phosphorylation
M) small KS
N) large KS
O) uncompetitive inhibition
P) [B]
R) A) and O)
Correct Answer

verified
L
Correct Answer
verified
Multiple Choice
At substrate concentrations much lower than the enzyme concentration,
A) the rate of reaction is expected to be inversely proportional to substrate concentration.
B) the rate of reaction is expected to be directly proportional to substrate concentration.
C) first order enzyme kinetics are not observed.
D) the KM is lower.
E) the rate of reaction is independent of substrate concentration.
G) D) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
For a reaction A + B C, if the concentration of B is much larger than A so that [B] remains constant during the reaction while [A] is varied, the kinetics will be
A) sigmoidal.
B) pseudo-first-order.
C) unimolecular.
D) zero-order.
E) hyperbolic.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Protein kinases are involved in
A) the digestion of drugs to potentially toxic byproducts.
B) the degradation of enzymes to the component amino acids.
C) the phosphorylation of a wide variety of proteins.
D) the metabolism of drugs to water soluble, excretable compounds.
E) all of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the figures below, which of the following expressions would be correct? 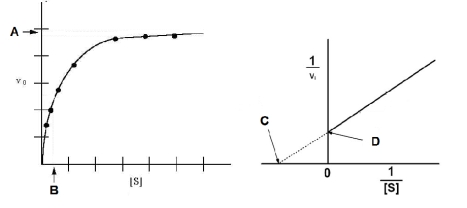
A) Vmax = 1/B
B) C = 1/ Vmax
C) D= Vmax
D) D = 1/ Vmax
E) A = 1/ Vmax
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Allosteric activators
A) bind via covalent attachment.
B) stabilize conformations with higher Ks.
C) stabilize conformations with higher substrate affinity.
D) all of the above
E) none of the above.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order for an enzymatic reaction obeying the Michaelis-Menten equation to reach 3/4 of its maximum velocity,
A) [S] would need to be equal to KM
B) [S] would need to be ½ KM
C) [S] would need to be 3KM
D) [S] would need to be ¾ KM
E) not enough information is given to make this calculation
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Matching -In uncompetitive inhibition, the inhibitor binds only to the ______.
A) isozymes
B) [A]
C) the rate constant
D) Ping Pong
E) bimolecular
F) ES complex
G) random ordered
H) competitive inhibition
I) unimolecular
J) [A]²
K) Competitive inhibition
L) phosphorylation
M) small KS
N) large KS
O) uncompetitive inhibition
P) [B]
R) H) and O)
Correct Answer

verified
F
Correct Answer
verified
Multiple Choice
Matching -In ______, the inhibitor binds to a site involved in both substrate binding and catalysis.
A) isozymes
B) [A]
C) the rate constant
D) Ping Pong
E) bimolecular
F) ES complex
G) random ordered
H) competitive inhibition
I) unimolecular
J) [A]²
K) Competitive inhibition
L) phosphorylation
M) small KS
N) large KS
O) uncompetitive inhibition
P) [B]
R) E) and M)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An extremely efficient enzyme called "efficase" catalyzes the conversion of "A" to "B." A researcher decides to mutate the enzyme in order to try to improve its performance.Following active site mutations, a significant reduction in the value of KM and Vmax was observed.Which of the following may have occurred?
A) The affinity of the enzyme for the substrate was increased to a point which did not favor propagation (continuation) of the reaction.
B) The decrease in Vmax was not related to the decrease in KM.
C) If the reaction was first-order, the change in KM cannot have affected Vmax.
D) The stability of E+S (E+A as written above) was increased, thereby increasing the KM.
E) The reverse reaction (breakdown of EA to E+A) was favored, slowing the Vmax.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A new drug has been discovered which inhibits the reaction catalyzed by enzyme A.Based on the information shown below, what is this drug? 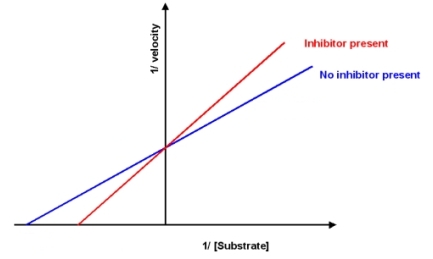
A) competitive inhibitor
B) uncompetitive inhibitor
C) mixed inhibitor
D) allosteric activator
E) More information is required to answer the question.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the initial velocity for an enzymatic reaction when Vmax = 6.5 × 10-5 mol•sec-1, [S] = 3.0 × 10-3 M, KM = 4.5 × 10-3 M and the enzyme concentration at time zero is 1.5 × 10-2 M.
A) 3.9 × 10-5 mol•sec-1
B) 2.6 × 10-5 mol•sec-1
C) 1.4 × 10-2 mol•sec-1
D) 8.7 × 10-3 mol•sec-1
E) Not enough information is given to make this calculation.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Parallel lines on a Lineweaver-Burk plot indicate I.an increase in KM. II.decrease in KM. III.decrease in Vmax. IV.uncompetitive inhibition.
A) I, IV
B) II, III, IV
C) I or II, III
D) I or III, II
E) I, III, IV
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Compounds that function as "mixed inhibitors" I.interfere with substrate binding to the enzyme. II.bind to the enzyme reversibly. III.can bind to the enzyme/substrate complex.
A) I
B) II
C) III
D) II, III
E) I, II, III
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following data were collected under conditions indicated in the graph below during the time period of 0-5 seconds.Upon plotting the Lineweaver-Burk plot, the information given in the table below was determined.Based on this available information which of the following is FALSE?
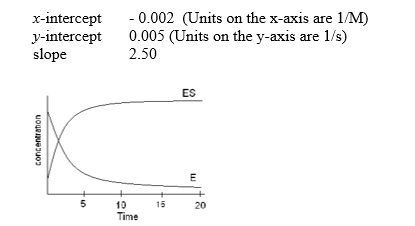
A) The Vmax equals 200 M/s
B) The Ks equals 500 M
C) The kapp equals 200 per second
D) The data was collected prior to reaching steady state.
E) The kcat cannot be determined for this information.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 55
Related Exams